A 2025 scoping review titled ‘Stroke at the Hairdresser’s Chair’, which was published in the American Journal of Emergency Medicine, synthesised data from 22 previous studies over five decades to provide a comprehensive look at Beauty Parlor Stroke Syndrome (BPSS), a rare but clinically significant phenomenon.
The review analysed 54 reported cases, finding that a significant majority (79.63%) were women, with occurrences linked to neck hyperextension during salon visits, dental procedures, and other activities. Researchers highlighted that while BPSS is uncommon, its symptoms can overlap with those of a classic stroke, emphasising the importance of thorough patient history and neuroimaging for an accurate and timely diagnosis. The study concluded that preventative strategies, such as providing neck support in salons and raising awareness among both clients and professionals, are essential for mitigating this risk.
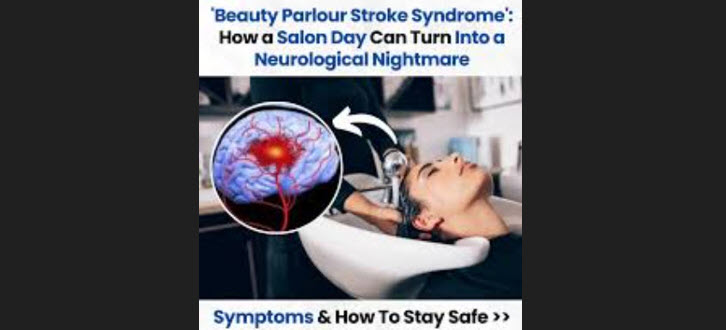
The term ‘Beauty Parlor Stroke Syndrome’, while it may seem colloquial, points to a clinically significant phenomenon: vertebral artery dissection or compression caused by sustained hyperextension of the neck. The setting of a salon shampoo bowl is the most commonly cited trigger, where a patient’s head is tilted backward over a basin’s edge for an extended period, creating mechanical stress on the vessels supplying the brain’s posterior circulation. First described by Dr. Michael Weintraub in 1993, the syndrome highlights how seemingly innocuous, everyday activities can precipitate serious neurological injury, particularly in susceptible individuals.
The underlying pathophysiology centres on compromise of the vertebral arteries, which ascend through the cervical spine to join and form the basilar artery. This critical system supplies blood to the brainstem, cerebellum, and posterior cerebrum. When the neck is hyperextended, especially against a hard surface, this can cause a vessel wall to dissect; a tear that can lead to intramural hematoma formation, luminal occlusion or the release of a clot that embolises to the brain. In other cases, external compression from osteophytes or soft tissues can temporarily restrict blood flow, a phenomenon known as vertebrobasilar insufficiency. The resulting posterior circulation strokes can manifest with symptoms such as dizziness, vertigo, ataxia, visual disturbances, and slurred speech, with symptom onset often delayed for hours or even days.
Epidemiological data, predominantly from case reports and small case series, indicates that while the syndrome is rare, it is disproportionately reported in women. For example, a recent scoping review identified that 79.63% of the 54 cases reviewed were female, with a wide age range from teenagers to the elderly. These findings are critical, as they underscore the need for awareness among patients and salon professionals alike. Risk factors for cervical artery dissection extend beyond the salon, encompassing other activities involving neck hyperextension, such as yoga, painting ceilings, or sudden movements like sneezing. Individuals with pre-existing vascular conditions, such as atherosclerosis, fibromuscular dysplasia or connective tissue disorders, may be at a heightened risk, though the syndrome can also affect young, otherwise healthy individuals.
Raising awareness in the beauty and wellness industries is a crucial preventative measure. Salons can mitigate risk by providing proper neck support, such as rolled towels or cushions, and adjusting chairs to reduce the degree of cervical hyperextension. Patients should be encouraged to communicate any discomfort or neck pain immediately during a shampoo. While the overall risk remains low, the potential for devastating long-term disability; from permanent balance issues to severe neurological deficits… warrants increased vigilance. For clinicians, maintaining a high index of suspicion is essential, particularly when evaluating patients presenting with posterior circulation symptoms following recent neck manipulation. A comprehensive patient history that includes recent activities is vital for accurate and timely diagnosis through neuroimaging. Ultimately, the phenomenon of ‘Beauty Parlour Stroke Syndrome’ is not an urban legend but a clinically confirmed, though rare, condition that necessitates proactive awareness and preventative strategies.